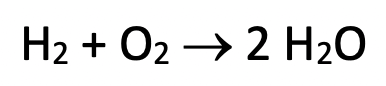BILANCIAMENTO REAZIONI REDOX 1) H2O2 + MnO4 - → Mn2+ + H2O + O2 ambiente acido 2) MnO4 2- → Mn2+ + CO2 ambiente acid
REGOLE PER L'ATTRIBUZIONE DEI COEFFICIENTI STECHIOMETRICI DI UNA REAZIONE OSSIDORIDUTTIVA SECONDO IL METODO DELLO IONE-ELETTRION

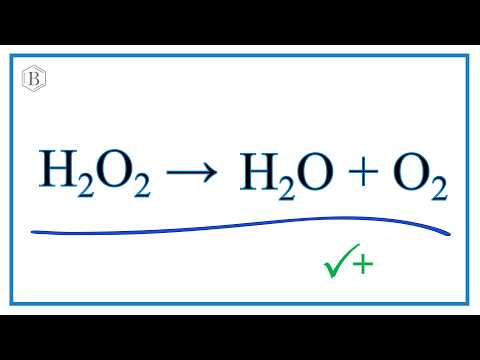
H2O+O2=H2O2 Balance the chemical equation. h2o+o2=h2o2 water and oxygen to form hydrogen peroxide - YouTube